AcceleDev places a high value on our R&D and production site management systems. We are committed to protecting the well-being of the environment as well as the health and safety of all our employees. AcceleDev’s EHS management system is established based on the best practices of the pharmaceutical industry. We have designed and developed state of the art facilities and safety protocols to ensure that EHS principles are a key component of our business operations which include:
Integrated safety and health management system (OHSAS18001)
Environment management system (ISO14001)
Process safety management (PSM)
We conduct process safety evaluations for all processes and utilize DSC and Sedex equipment to facilitate in-depth HAZOP testing and reviews. We have passed a number of client audits from top pharmaceutical and biotech companies and third party authorities
All wastewater from our labs and production facilities is captured, analyzed, and treated according to strict site and government regulations. Solid waste and waste gases are collected, monitored, treated and disposed of according to strict site and government regulations/ oversight.
We have a well-established Quality Management system in place which includes comprehensive SOP protocols and an extensive employee training program. SOPs are developed based on ICH Q7, covering the entire quality system at manufacturing sites.
We have a well-defined Materials Management system, including a dedicated raw material storage area and controlled environment (temperature and humidity) storage facility for the products we produce.
AcceleDev has been recognized for our commitment to EHS and was awarded a “Silver Recognition Level” for Corporate Social Responsibility (CSR) for sustainable supply management by EcoVadis, an independent leader in business sustainability assessment. AcceleDev received an overall score in the 67% percentile of the companies rated by EcoVadis. AcceleDev has been a Ecovadis Silver award winner, maintaining high standards for 7 consecutive years.
Our sites have successfully completed audits by a number of large US and European pharmaceutical companies.
One of our recent audit reports summarized that “AcceleDev QC department is knowledgeable, cooperative, sincere and honest” and “AcceleDev R&D operates with full transparency”.
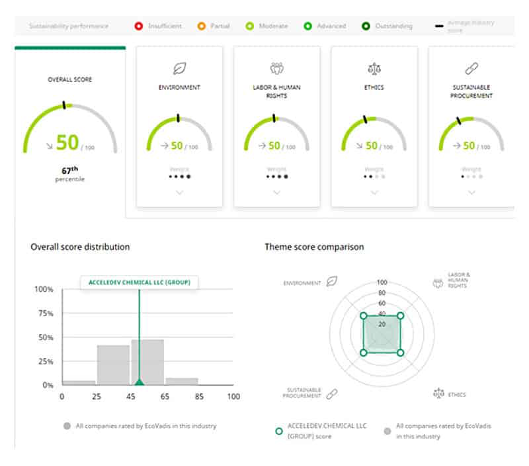
The EcoVadis ( www.ecovadis.com ) methodology is at the core of our CSR analysis system, covering 21 criteria across four themes of Environment, Fair labor & human rights, ethics, and sustainable procurement.
The methodology is built on international CSR standards including the Global Reporting Initiative, the United Nations Global Compact, and the ISO 26000, covering 190 spend categories and 150 countries.
